3D illustration displaying mind and energetic vagus nerve (tenth cranial nerve or CN X)
Supply: Axel_Kock/Shutterstock
Stimulating the vagus nerve prompts the parasympathetic department of the autonomic nervous system in ways in which curb fight-or-flight stress responses, which has a relaxing impact on the physique and thoughts.
By counteracting hyperarousal and the sympathetic nervous system’s stress response, vagus nerve stimulation can also cut back post-traumatic stress dysfunction (PTSD) signs by curbing inflammatory responses.
A latest pilot research (Bremner et al., 2021) of a non-invasive transcutaneous cervical vagal nerve stimulation machine discovered that three months of tcVNS therapy “resulted in a 31% larger discount in PTSD signs in comparison with sham therapy.”
“These preliminary outcomes recommend that tcVNS reduces inflammatory responses to emphasize, which can partly underlie useful results on PTSD signs,” the authors clarify. Omer Inan of the Georgia Institute of Expertise’s Faculty of Electrical and Laptop Engineering and the Coulter Division of Biomedical Engineering was this research’s senior creator.
This randomized, double-blind research was revealed in December of 2021 within the peer-reviewed Journal of Affective Issues Reviews. This analysis corroborates different sham-controlled analysis (Bremner et al., 2020) by the identical crew, which discovered that stimulating the vagus nerve utilizing a handheld tcVNS machine curbs inflammatory responses to emphasize as listed by pro-inflammatory interleukin-6 (IL-6) cytokine biomarkers.
One other preliminary research (Lamb et al., 2017) of a non-invasive VNS machine that targets the auricular department of the vagus nerve by way of the outer ear discovered that this machine elevated parasympathetic exercise and decreased sympathetic nervous system exercise, which had optimistic results on hyperarousal and the autonomic state of sufferers with PTSD. (See, “Vagus Nerve Stimulation via the Outer Ear Takes Center Stage.”)
“These results recommend that tVNS could modulate emotional state as mirrored by downregulating fight-or-flight and upregulating a physiological state conducive to optimistic social engagement,” first creator Damon Lamb of the College of Florida and co-authors write.
The entire abovementioned analysis into non-invasive vagus nerve stimulation as a doable therapy for PTSD is in its early phases. These preliminary pilot research had been small; extra analysis involving a bigger pattern dimension is required.
Nonetheless, there’s a sense of urgency to assist fight veterans and others with PTSD who have not benefited from conventional therapies by giving them doctor-supervised entry to new “breakthrough” therapies. Due to this fact, the U.S. Meals and Drug Administration (FDA) has fast-tracked a few of these units by way of their “Breakthrough Devices Program.”
What Is the FDA’s Breakthrough Units Program?
As a part of the FDA’s mission to “shield and promote public well being,” they provide producers an expedited pathway to supply sufferers and well being care suppliers quicker entry to lately invented medical units by dashing up how these breakthrough units are developed, assessed, and reviewed.
The Breakthrough Units Program facilitates direct communication between the producers of pioneering bioelectronic units and folks on the FDA in the course of the premarket evaluation part. By getting the FDA’s suggestions a couple of novel medical machine early on, producers can deal with potential issues in a well timed approach whereas preserving the statutory requirements for premarket approval, De Novo classification requests, and 510(k) clearances.
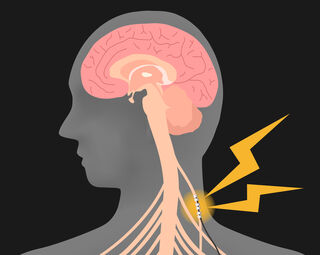
Non-Invasive vagus nerve stimulation (nVNS) doesn’t require surgical procedure or a completely implanted machine. Non-invasive VNS bioelectronic units ship a doctor-prescribed dosage of electrical energy that penetrates the pores and skin and targets the vagus nerve on the facet of a affected person’s neck (tcVNS) or by way of the outer ear (taVNS).
Supply: metamorworks/Shutterstock
Two Non-Invasive VNS Units Have been Just lately Given “Breakthrough System Designation” by the FDA for PTSD Therapy
In July 2021, a non-invasive earbud machine known as the Phoenix—which delivers a gentle electrical tingle that stimulates the vagus nerve by way of a affected person’s outer ear—received Breakthrough Device Designation from the FDA to deal with post-traumatic stress dysfunction. This transcutaneous auricular vagal nerve stimulation (taVNS) machine addresses underlying autonomic nervous system imbalances related to PTSD by growing parasympathetic exercise.
Publish-Traumatic Stress Dysfunction Important Reads
In January 2022, a transcutaneous cervical vagal nerve stimulation (tcVNS) machine known as gammaCORE additionally acquired FDA Breakthrough System Designation to deal with PTSD. This handheld nVNS machine stimulates the vagus nerve with pulses of electrical energy delivered by way of the pores and skin on the facet of a affected person’s neck.
“Present therapies for post-traumatic stress dysfunction involving medication and psychotherapy have limitations attributable to restricted efficacy, doable unwanted effects, and the unwillingness of many PTSD sufferers to have interaction in therapies that contain reliving traumatic reminiscences,” J. Douglas Bremner of the Emory College Faculty of Medication stated in a news release about gammaCORE being a part of the FDA’s Breakthrough Units Program.
“GammaCORE [non-invasive tcVNS] represents a brand new class of therapy separate from remedy or psychotherapy that’s secure, comparatively freed from unwanted effects, and doesn’t contain pricey and invasive procedures for implantation, like earlier VNS units permitted by the FDA for therapy of refractory depression,” he added.
These novel applied sciences are promising and provide hope, however the FDA hasn’t but permitted these nVNS units for PTSD therapy. As talked about, extra analysis is required.
Notably, a latest important evaluation (Yap et al., 2020) of tVNS revealed by Frontiers in Neuroscience concludes: “Extra rigorous systematic research are required to research the consequences of stimulation parameters, websites of stimulation, and electrode varieties on mind activation and medical outcomes.”
Till non-invasive vagus nerve stimulation will get its official seal of approval from the FDA for treating post-traumatic stress dysfunction, it is in all probability wise to proceed with warning and never overhype the potential of those units.
Battle of Curiosity Assertion: This text just isn’t an endorsement of the Phoenix or gammaCORE non-invasive vagus nerve stimulation (nVNS) units, neither is this reportage on their Breakthrough System Designation for the therapy of PTSD by the U.S. Meals and Drug Administration supposed as medical recommendation or a advice. This publish’s creator just isn’t paid by or related to electroCore Inc. or Evren Applied sciences, Inc.; there isn’t any battle of curiosity.
As all the time, ask your physician in regards to the doable advantages and potential dangers of utilizing any bioelectronic medical machine and comply with the prescribed utilization directions carefully.